Validation and development of a turbulence-chemistry interaction model by Direct Numerical Simulation database
Shaowu Pan, Yun Bai
State Key Laboratory of Clean Energy Utilization
Zhejiang University, Hangzhou, China
Advised By Professor kun Luo
Overview
A promising model in for turbulence-chemistry interaction is proposed a few years ago but no detailed validation by DNS data existed. DNS data of a spray combustion of heptane and air in a cube box with 6.28 mm in each direction, was taken as an example. Chemical kinetics is restricted to one-step global reaction since the purpose here is to evaluate the global behavior of spray combustion without knowing the reaction details. This research so far has been divided into three parts . First, evaluation of the applicability of this model is performed. Previous study on validation of the second-order moment combustion model by DNS didn't consider the effect of compressibility Due to the fact that density varies greatly in real combustion, it may induce error in simulation of combustion. Moreover, unity order chemical reaction mechanism (unity power of concentration in Arrhenius equation) was assumed in original theory, while most reactions are non-unity .Second, necessary modifications for this model are proposed in order to meet the configuration of available DNS data. It contains both consideration for compressibility and non unity order chemical reaction mechanism. Third, validation on the assumptions in the modelling process is conduct to instruct combustion modelling . (figure above illustrates an iso-surface of moderate mixture fraction in one piece of 16 computational regions)
A promising model in for turbulence-chemistry interaction is proposed a few years ago but no detailed validation by DNS data existed. DNS data of a spray combustion of heptane and air in a cube box with 6.28 mm in each direction, was taken as an example. Chemical kinetics is restricted to one-step global reaction since the purpose here is to evaluate the global behavior of spray combustion without knowing the reaction details. This research so far has been divided into three parts . First, evaluation of the applicability of this model is performed. Previous study on validation of the second-order moment combustion model by DNS didn't consider the effect of compressibility Due to the fact that density varies greatly in real combustion, it may induce error in simulation of combustion. Moreover, unity order chemical reaction mechanism (unity power of concentration in Arrhenius equation) was assumed in original theory, while most reactions are non-unity .Second, necessary modifications for this model are proposed in order to meet the configuration of available DNS data. It contains both consideration for compressibility and non unity order chemical reaction mechanism. Third, validation on the assumptions in the modelling process is conduct to instruct combustion modelling . (figure above illustrates an iso-surface of moderate mixture fraction in one piece of 16 computational regions)
|
|
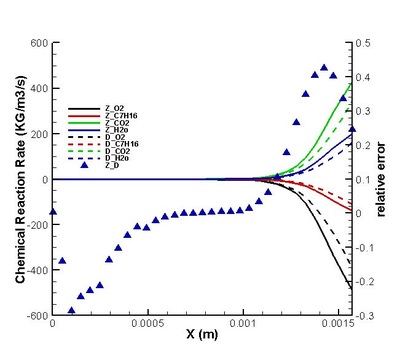
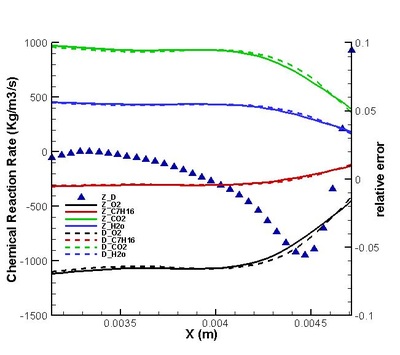
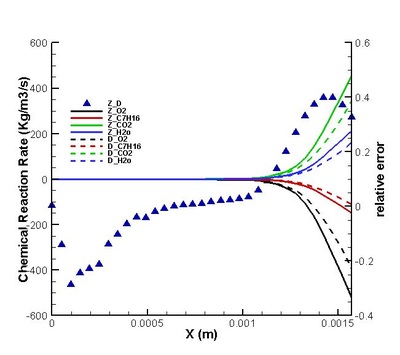
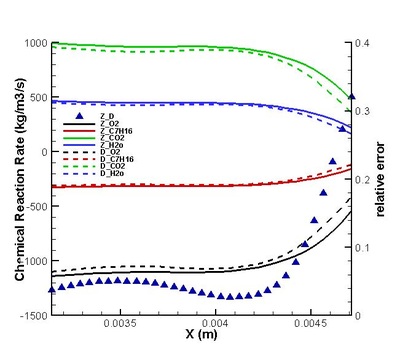
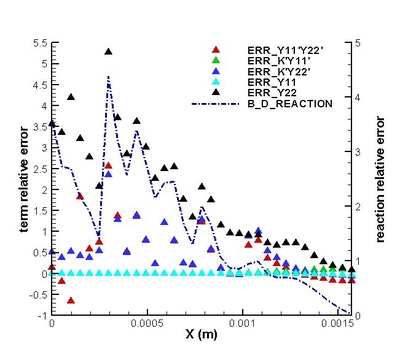
Validation on modifications of original model was conduct. The regions for strong and weak reaction can be defined by comparing the chemical reaction rate distribution streamwise .Moreover, the DNS statistical results shows that the omission of third order correlation in original theory is valid in strong reaction region but would induce error in weak reaction region (see figure on the left). That two-delta PDF closure of time-averaged temperature exponential term is perfect except in the interface between strong and weak reaction region. (see figure on the left) It suggests further attention on modifications should be paid to this interface. Moreover, the variation of density leads to a moderate deviation in reaction rate but negligible in strong reaction region. (see figures below)
|
|
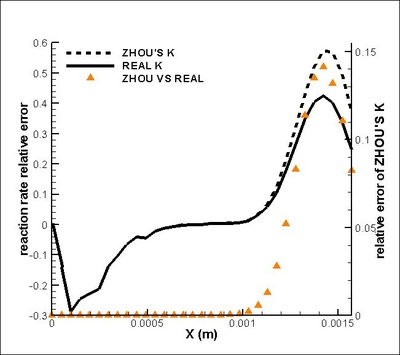
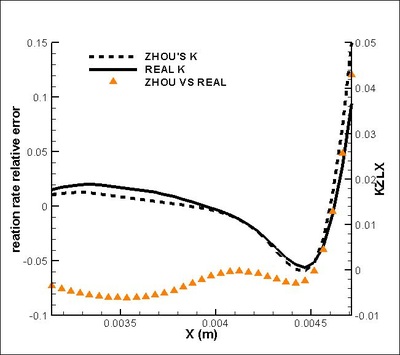
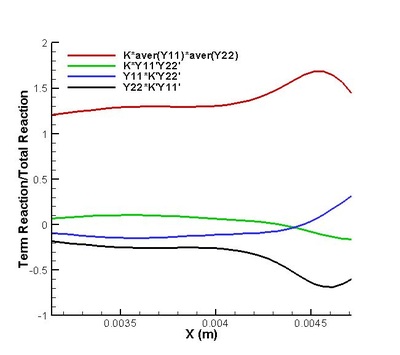
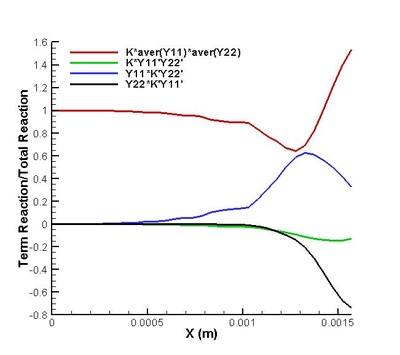
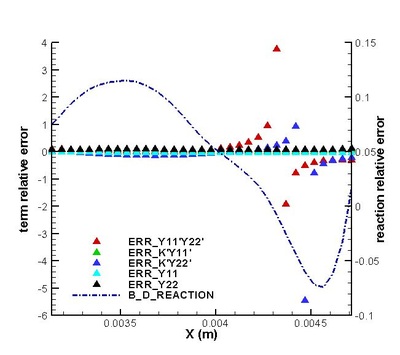
It is found that the averaged term contributed the most to the chemical reaction rate. Among all the second order correlation terms, the contributions of concentration-concentration terms could be neglected in both strong and weak reaction region in the case discussed. But the contri- butions of temperature-concentration correlation can not be neglected in certain areas, which appears to be strong negative in the interface between strong and weak reaction region. Another important finding that should be noted is that in most places in strong reaction region, the averaged term of reaction rate is higher than actual value. Next, validations on modifications are conduct. It is found that assumptions for Y11'Y22', K'Y11, K'Y22', Y11, Y22 (all specified terms in modification) are satisfactory in strong reaction region but poor in weak reaction region. (see figure on the right) The resulted relative error in chemical reaction rate was within 12% in strong reaction region except a great deviation existing at a distance off the boundary.
|
|
To be continued